Clinical Evidence
Effect of Continuous Glucose Monitoring on Hypoglycemia in Older Adults with Type 1 Diabetes: A Randomized Clinical Trial
Published on: 9 November 2021
The Wireless Innovation for Seniors With Diabetes Mellitus (WISDM) study, published in the Journal of American Medical Association in 2020,1 was one of the first randomized control trials to assess whether Dexcom CGM use was more effective in reducing hypoglycemia than self-monitoring of blood glucose (SMBG) in older adults with type 1 diabetes (n=203, >60 years of age). Compared with the SMBG control group, the Dexcom CGM group achieved the following results:
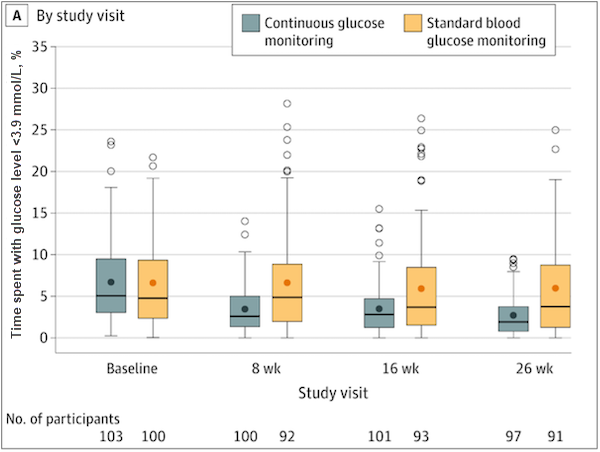
- Median percent time <70 mg/dL decreased from 5.1% (73 minutes per day) at baseline to 2.7% (39 minutes per day) at the 6-month follow-up for the CGM group. By contrast, hypoglycemia exposure remained relatively unchanged for the SMBG group—from 4.7% (68 minutes per day) at baseline to 4.9% (70 minutes per day) during follow-up—for an adjusted treatment group difference of −1.9% (95% CI, −2.8% to −1.1%; P <0.001).
- This corresponds to a significant reduction in hypoglycemia of 27 minutes per day (P <.001).
- Reduction in hypoglycemia observed in the CGM group was independent of baseline characteristics, including age (<70 vs ≥70 years), socioeconomic status, presence of cognitive impairment, or A1C.
- Mean time spent in the target range of 70 to 180 mg/dL was 8.8 percentage points (2.1 hours per day) higher in the CGM group compared with the SMBG group (P <0.001).
- Baseline A1C was relatively low. However, the CGM group observed a small but statistically significant decrease in mean A1C (adjusted group difference, −0.3%; P <0.001).
- Adherence to CGM was high; 83% of users wore their Dexcom CGM >6 days per week.
Percent of time spent in hypoglycemia (<3.9 mmol/L) by study visit
1. Pratley RE, Kanapka LG, Rickels MR, et al. Effect of Continuous Glucose Monitoring on Hypoglycemia in Older Adults With Type 1 Diabetes: A Randomized Clinical Trial. JAMA. 2020;323(23):2397–2406.
LBL022291 Rev001
